The full film, not isolated snapshots.
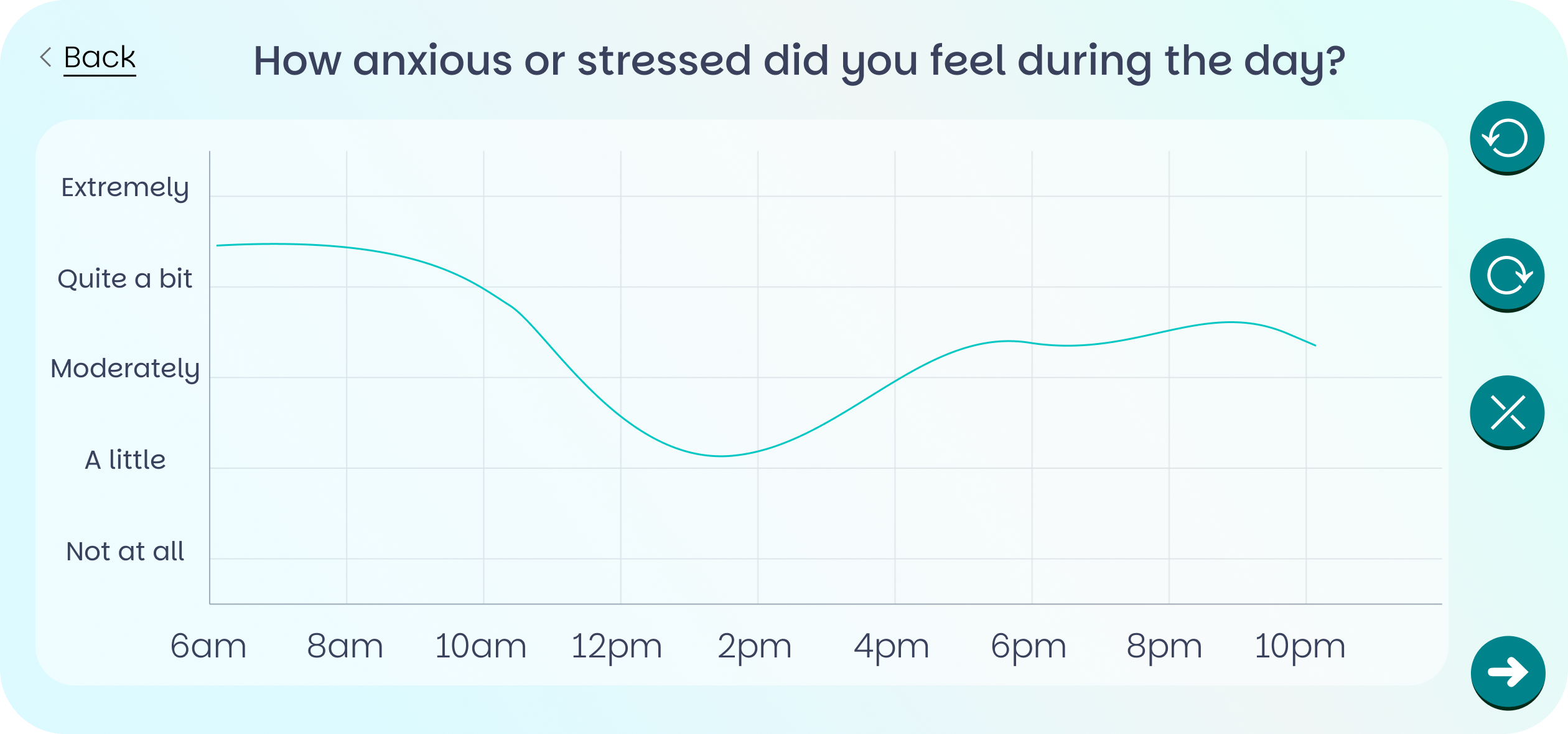
Traces (Temporal Experience Tracing™) is a validated data collection method that captures how patients experience their symptoms continuously throughout each day - not as a single retrospective score, but as a rich time-series that mirrors the natural rhythm of lived experience.
Each evening, patients draw a simple continuous curve on their phone or tablet showing how their symptom fluctuated across the day. It takes under two minutes. The result is 100× more data per participant than standard questionnaires, with lower bias and measurably greater sensitivity to treatment effects.
TET data integrates seamlessly with EEG, wearable signals, sleep tracking, and activity monitoring - bridging the granularity gap between physiological data and the patient experience it's meant to reflect.






